文章信息 | Novel Iterative Genome Mining and Engineering of ABifunctional KvVDH Enable Selective Production of Furan Carboxylic Acids from High-Concentration 5-Hydroxymethylfurfural, Green Chemistry, 2026. | 作者 | Yanan Cui, Jie Zhang, Xijun Xing, Hanwen Zhang, Yutong Chen, Liqiang Fan, Xu Li, Yongjun Qiu, Chen Deng*, Liming Zhao* | 摘要 | Enzyme catalysis provides a promising strategy for the selective conversion of biomass-derived chemical 5-hydroxymethylfurfural (HMF) into value-added furan carboxylic acids. However, the enzymatic synthesis of 5-hydroxymethyl-2-furancarboxylic acid (HMFCA), 5-formyl-2-furancarboxylic acid (FFCA) and 2,5-furandicarboxylic acid (FDCA) faces two critical bottlenecks: scarcity of bifunctional enzymes active toward HMF, HMFCA and FFCA with low activity, as well as severe high-concentration HMF inhibition. In this study, to address these gaps and expand the enzymatic synthesis pathway of furan carboxylic acids, we developed a data-driven “molecular probe and function-mediated iterative genome mining” strategy (32.1% functional enzyme discovery rate, a threefold improvement over traditional homology screening) to identify 7 novel target enzymes from 13 HMF-tolerant strains and protein database. Among them, novel bifunctional vanillin dehydrogenase KvVDH exhibited the highest HMF activity (873.1 mU·mg⁻¹). Subsequently, we further engineered KvVDH via KnowVolution (computational design and directed evolution), obtaining the optimal variant M3 (A50S/Y370W) with 57.5-fold higher catalytic efficiency (kcat/Km = 5.75 vs 0.10 s⁻¹·mM⁻¹ for WT KvVDH), 68.8% residual activity at 40 °C and 1 h (15.6-fold vs WT KvVDH), and exceptional high-HMF tolerance: whole-cell catalysts achieved 97.8% HMFCA yield from 240 mM HMF. Structural and molecular dynamics analysis results indicated that the potential factors enhancing M3 (A50S/Y370W) activity and thermostability include the remodeling of the restructuring of the hydrogen bond network, conformational changes in substrate channel, regulation of the local conformation at the active site, and reduction in the binding free energy. Furthermore, coupling M3 (A50S/Y370W) with nicotinamide oxidase (NOX) to catalyze 90 mM HMF obtained FFCA with a yield as high as 98.6%. Based on these results, a one-pot two-step enzymatic process was established. Starting from 90 mM HMF, the final FDCA yield was as high as 95.3% through sequential catalysis by M3 (A50S/Y370W), NOX and EcALDH. This study develops an excellent enzymatic toolkit for the high efficiency and high selectivity conversion of HMF, laying the foundation for the green and economical production of furan carboxylic acids. | 摘要图 | 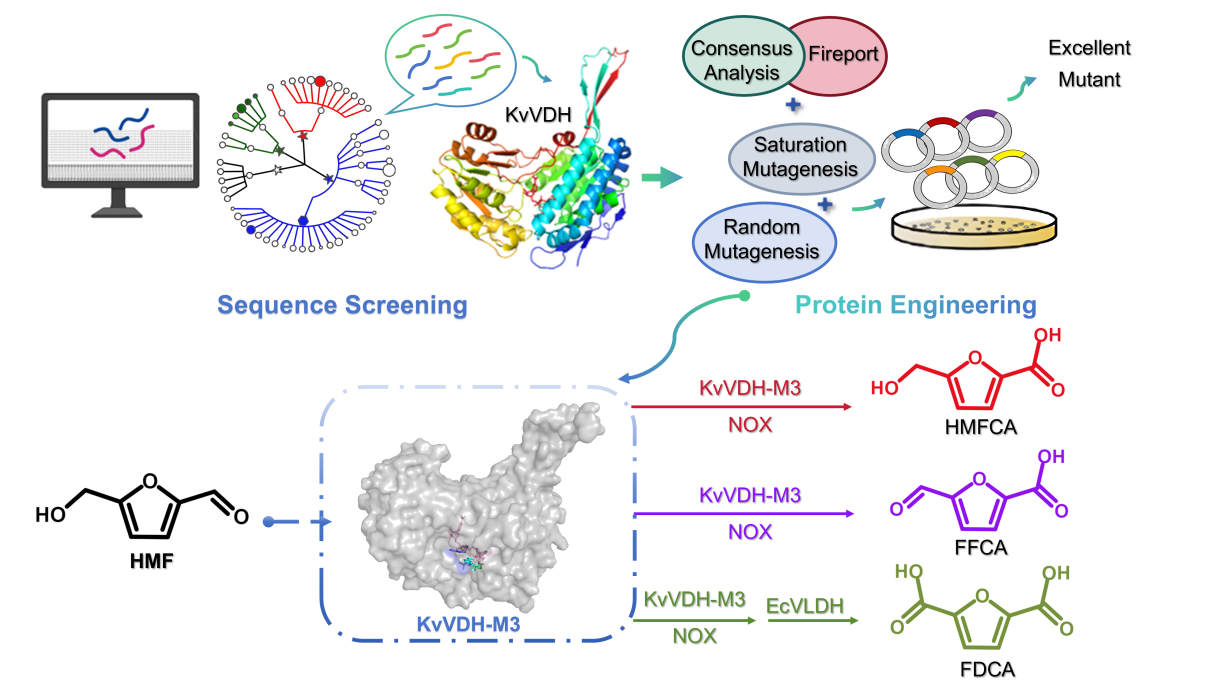
|
|